Egg Freezing
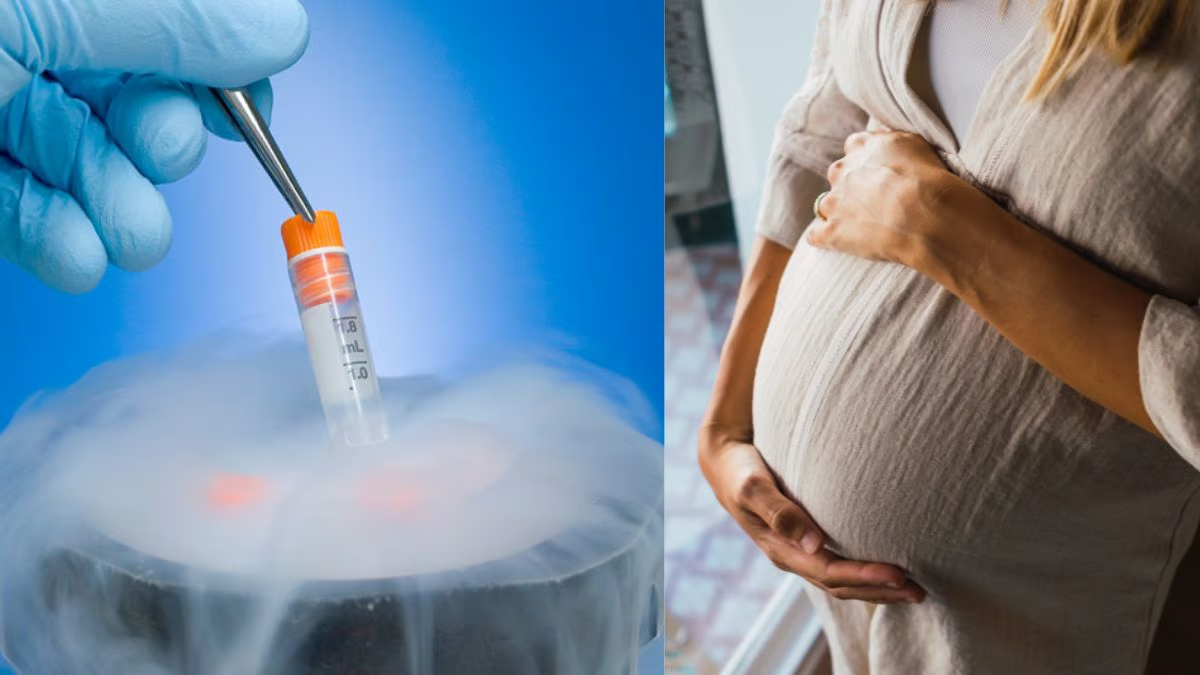
Egg Freezing: A Vision for Reproductive Autonomy
In an era where career aspirations and personal goals shape life’s timeline, egg freezing stands out as a beacon of hope for women aiming to balance their professional ambitions with the desire for future parenthood. Cryopreservation, the technique behind egg freezing, offers women the chance to preserve their fertility, enabling them to decide when they’re ready to embark on the journey of motherhood. The process is especially beneficial for younger women, where the vitality of eggs enhances the prospects of successful pregnancies post-thawing.
Who Benefits from Oocyte Freezing?
Career-focused Women
For those prioritizing professional growth and wishing to postpone pregnancy.
Cancer Patients
Women about to undergo treatments like chemotherapy or radiotherapy, which can impact fertility.
Risk of Premature Ovarian Failure
Individuals with a medical predisposition to losing ovarian function at an early age.
Ethical or Religious Considerations
Women who prefer not to freeze embryos due to personal beliefs.
Optimal Timing for Egg Freezing
The decision to freeze eggs is a deeply personal one, with the ideal timing being whenever a woman chooses to delay pregnancy for any reason. The earlier in life this choice is made, the better the chances for future success due to the higher quality of younger eggs.
Longevity of Frozen Eggs
Remarkably, there’s no definitive limit to how long eggs can remain frozen while retaining their potential for a successful pregnancy. Successful pregnancies have been achieved using eggs frozen for a decade or more, highlighting the technique’s reliability and the enduring nature of cryopreserved eggs.
Safety of Egg Freezing
Comprehensive studies, including one of the largest examining over 900 babies born from frozen eggs, confirm that egg freezing is safe. These studies show no significant differences in health risks compared to babies conceived naturally, reassuring women that freezing eggs does not compromise the health of future offspring.
The Indigo Women's Center Edg
At Indigo Women’s Center, we’re committed to empowering women with the freedom to plan their futures without compromise. Our state-of-the-art cryopreservation services not only ensure the safe and effective freezing of eggs but also offer comprehensive support throughout the process. Our expert team is dedicated to providing personalized care, addressing each woman’s unique needs and concerns, and ensuring the best possible outcomes for when the time comes to start a family.
Embrace the Future on Your Terms
Egg freezing at Indigo Women’s Center is more than a medical procedure; it’s a commitment to supporting women’s choices and enabling them to pursue their dreams with the confidence that their reproductive futures are secure. Whether you’re climbing the career ladder, undergoing medical treatment, or simply not ready for parenthood, egg freezing offers a path to motherhood that aligns with your timeline. Explore the possibilities with us, and take the first step towards securing your future family on your terms.

