About Us
Menu Home About Us Team Services Reproductive Medicine Laparoscopy Cosmetic Gynecology Maternity Specialization Fibroids Endometriosis

At Indigo Women’s Centre, an NABH Accredited and ISO certified facility, we are dedicated to providing world-class fertility and women’s health care in a compassionate and personalized manner. With over 35 years of pioneering work in fertility treatments, our team of highly skilled specialists is committed to delivering excellence in Reproductive Medicine, Advanced Laparoscopy, Cosmetic Gynecology, and Maternity Services.
Our comprehensive services include IUI, IVF/ICSI, PGT, and Cryofreezing, guided by a team of fertility specialists, embryologists, and radiologists working together to optimize your fertility potential
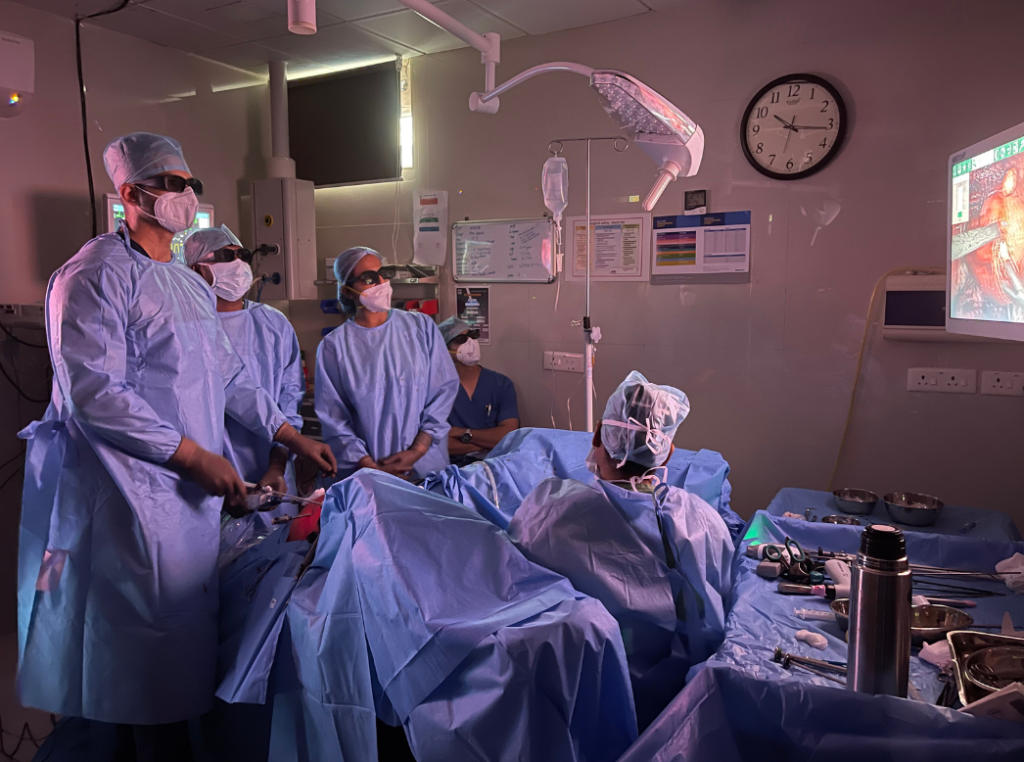
Equipped with state-of-the-art 3D laparoscopic units, we offer advanced procedures for fibroids, endometriosis, pelvic pain, and more, minimizing recovery time and enhancing outcomes.

Enhancing your well-being and confidence with procedures like Labioplasty, Vaginoplasty, Hymenoplasty, and the innovative G-Shot.

Experience a memorable journey to motherhood with our comprehensive care, from conception to delivery, in a comfortable and supportive environment.
Our "patients first" philosophy ensures you receive the highest quality care tailored to your needs, supported by the latest technology and evidence-based treatments.
Our specialists bring decades of experience and a track record of success to your treatment.
As pioneers in introducing 3D laparoscopy in Chennai, we offer precision and excellence in surgical treatments.
Our hospital’s strategy is“patients first”, where we give top priority to quality healthcare and patient satisfaction above everything else.
From one-to-one nursing care to personalized appointment scheduling, we prioritize your comfort and convenience at every step of your journey.
Fertility Solutions, Laparoscopic Procedures, and Cosmetic Gynaecology: All Under One Roof
Our "patients first" philosophy ensures you receive the highest quality care tailored to your needs, supported by the latest technology and evidence-based treatments.
Our specialists bring decades of experience and a track record of success to your treatment.
As pioneers in introducing 3D laparoscopy in Chennai, we offer precision and excellence in surgical treatments.
Our hospital’s strategy is“patients first”, where we give top priority to quality healthcare and patient satisfaction above everything else.
From one-to-one nursing care to personalized appointment scheduling, we prioritize your comfort and convenience at every step of your journey.

DIRECTOR OF REPRODUCTIVE MEDICINE

DIRECTOR OF LAPAROSCOPIC SURGERY

CONSULTANT RADIOLOGIST

CONSULTANT PAEDIATRICIAN AND NEONATOLOGIST
Book an appointment to start your journey with Indigo Women’s Centre, where we promise a fruitful path to parenthood. Explore our services, meet our team, and read testimonials from our many happy families.
Yes, we are here to help you start your journey, and we promise it will be a fruitful one!